EXACTECH RECALL
Medical Device Lawyer investigating recalled Exactech knee implants for injured plaintiffs nationwide
Exactech Knee Implant Recall
Investigating recalled Exactech knee and ankle replacements
Reviewing Exactech Recall claims for patients implanted with Optetrak, Truliant and Vantage devices. Compensation may be available. Contact The Lyon Firm for a free consultation.
Between the years of 2004 and 2022, thousands of total knee replacement systems were manufactured by Exactech, many of which could fail prematurely, according to the company.
Exactech learned that one of the packaging layers for the plastic implant insert has been out of specification and may allow oxygen from the air to diffuse into the plastic insert prior to it being implanted in a patient. Resulting oxidation can cause the plastic to deteriorate earlier than expected or to become damaged after it is implanted.
Premature wear of the plastic insert of a knee replacement can lead to the need for revision surgery. Exactech is reportedly assisting patients with some out-of-pocket costs related to additional surgery that may be necessary, but it is best to speak to an attorney before accepting any assistance.
Millions of patients have been implanted with Knee replacement systems since the devices were brought to market. However, design issues with the Exactech Optetrak knee replacement system and other knee implants have prompted the companies and the U.S. Food and Drug Administration (FDA) to issue warnings and recalls for the potentially dangerous product.
Thousands of patients have experienced problems with the Exactech Optetrak, Exactech Truliant and Exactech Vantage, including pain and tibial loosening, a condition which may require patients to undergo a painful revision surgery to resolve implant complications.
Attorneys representing injured patients note that the FDA’s adverse event reporting database had received over 1,000 reports of knee implant failures, many of which have resulted in knee revision surgeries.
The FDA has issued recalls after receiving numerous reports of knee implant tibial loosening. The FDA warned that knee tibial loosening is caused by the tibial baseplate loosening, which may lead to infection and inflammation in the surrounding bone and muscle. Those who have suffered from this device may have viable Exactech Optetrak Recall Injury Lawsuits.
Recalled Knee & Ankle Replacements
- OPTETRAK® All-polyethylene PS Tibial Components
- OPTETRAK® CR Tibial Inserts
- OPTETRAK® CR Slope Tibial Inserts
- OPTETRAK® PS Tibial Inserts
- OPTETRAK® HI-FLEX® PS Tibial Inserts
- OPTETRAK Logic®
- OPTETRAK Logic® CR Slope Tibial Inserts
- OPTETRAK Logic® CRC Tibial Inserts
- OPTETRAK Logic® PS Tibial Inserts
- OPTETRAK Logic® PSC Tibial Inserts
- OPTETRAK Logic® CC Tibial Inserts
- TRULIANT® CR Slope Tibial Inserts
- TRULIANT® CRC Tibial Inserts
- TRULIANT® PS Tibial Inserts
- TRULIANT® PSC Tibial Inserts
- VANTAGE® VANTAGE® Fixed-Bearing Liner
Joe Lyon is a highly-rated medical device recall lawyer and product liability attorney representing plaintiffs nationwide in a wide variety of defective medical device and civil litigation claims and Exactech Recall lawsuits.
CONTACT THE LYON FIRM TODAY
Please complete the form below for a FREE consultation.
Exactech Recall & Replacement Failure
Exactech has expanded the recall to include all knee and ankle arthroplasty polyethylene inserts packaged in non-conforming bags regardless of label or shelf life. During the period between August 2021 and January 2022, thousands of non-conforming knee and ankle devices have been shipped and implanted by surgeons.
There have been approximately 147,700 inserts implanted in the U.S. since 2004 that were produced with non-conforming packaging.
Three generations of Exactech knee systems have had polyethylene inserts packaged in non-conforming bags at various points during their respective market presence. The original Optetrak Knee system, from 1992, has shown higher overall revision rates.
Exactech ankle arthroplasty systems, or the Vantage® Total Ankle system, has been recalled for the same reasons as the knees. For patients who exhibit premature polyethylene wear, surgeons can consider revision surgery.
FDA has classified this field action as a class II recall, meaning that exposure to the product may cause medically reversable health consequences.
Many knee implant operations are successful and the devices function well for decades. But that is not always the case. Knee implant failure can be the result of a defectively designed component.
In the case of the Exactech Optetrak device, many reports indicate a loosening of the tibial tray, one of the device’s components linked to potential device failure.
The Optetrak component was deemed prone to excessive wear by the FDA and subject to a Class 2 device recall. Some patients who have received the Exactech implant have needed revision surgery to remove and replace the failed knee implant. Defective implants may not adhere fully and correctly to the bone, and lead to loosening, instability, pain, and infections.
Medical Device Recall & Injury
A study published in the medical journal Orthopaedics & Traumatology: Surgery & Research analyzed patients who were implanted with an Exactech total knee replacement system.
After about two years, the researchers found that tibial loosening was a common occurrence in the cement-tibial-implant component of the device, resulting in fragmentation. The implant failure was reportedly caused by tibial insert wear and tear of the implant.
Around 15 percent of patients said they were disappointed or dissatisfied with the implant. Over 20 percent of patients complained of Exactech knee pain that requires regular painkillers.
Around 22 percent were beginning to suffer from tibial implant loosening at only 25 months. Over a dozen of the reviewed patients needed revision surgery for tibial loosening, instability or pain. Revision surgery has its own risks of complications and infection.
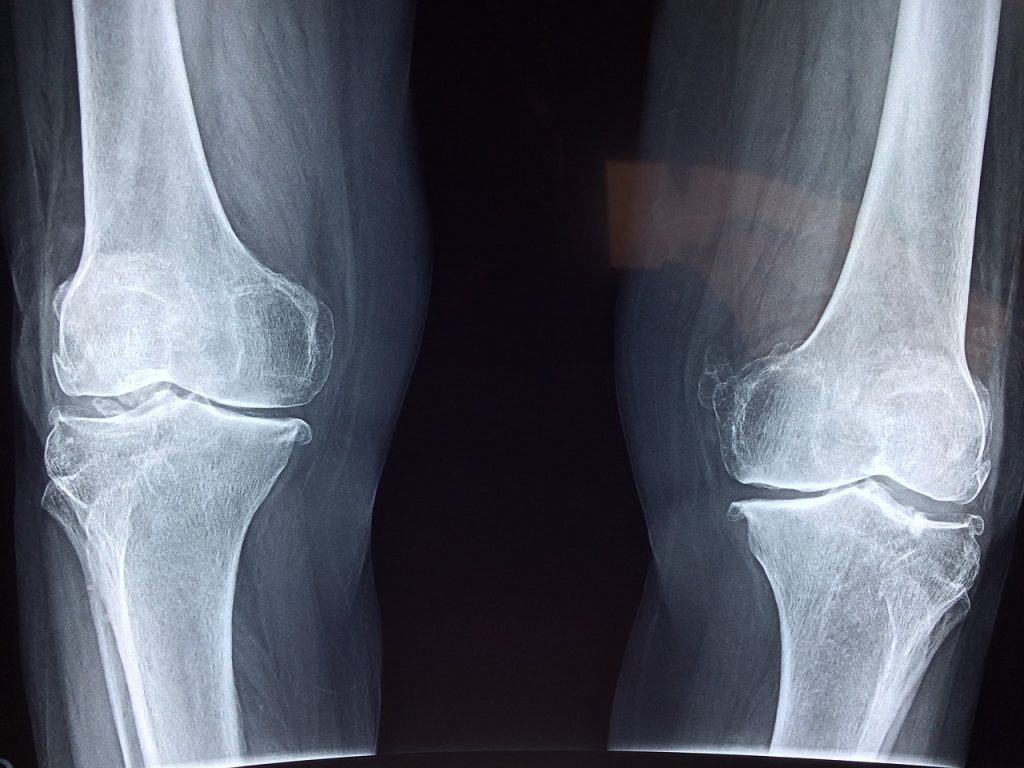
Exactech Knee Implant Recalls
The most recent extended Exactech recall involves knee and ankle implants, including the following models:
- Exactech Optetrak
- Exactech Truliant
- Exactech Vantage
A February 2022 Exactech implant recall was extended from an earlier 2021 recall due to improper packaging that allegedly impacted the safety and performance of the implants.
A study published in June, 2016, in the Journal of Arthroplasty concluded that some Knee implants may be associated with a high rate of baseplate loosening and failures that increase the risk of bone loss.
A related study published in the Journal of Knee Surgery said after reviewing patient and doctor reports, hospital databases, as well as independent studies, researchers encountered a high rate of debonding of the tibial implant–cement interface.
Patients with Exactech implants presented with pain on weight bearing and a decreased range of motion within 2 years after surgery. Numerous tibial failures were reported though researchers believed complications were underreported due to failure of radiographs to assess loosening.
Exactech Recall Lawsuits
When Exactech marketed their Optetrak product, the company promised patients a quick recovery time and a device manufactured to sustain harsh movements like exercise. Exactech has said the device was supposed to last longer and perform better than other knee implants on the market.
But many of the devices sold and implanted have been defective, and Exactech products have been causing further harm and injuries as a result. Patients who received Exactech knee implants were not expecting to undergo revision surgery for more than 15 years, making their early device failures a source of great distress and an important medical and legal matter. Complications associated with Exactech knee replacements include:
- Failure to bond
- Components wearing
- Fracture
- Implant detached from bone
- Persistent pain
- Bone Loss
- Infection
- Decreased range of motion
- Tissue Damage
- Nerve Damage
- Metallosis—metal debris shed into bloodstream
- Revision Surgery

Why are these cases important?
Defective medical device litigation helps to improve patient safety and holds medical device companies accountable when defective products injure plaintiffs and clients.
Companies may voluntarily recall their products, but the fact remains that much damage has already been done by the time some defective implants are recalled. Companies have a responsibility to properly test their devices before they are released to the public.
CONTACT THE LYON FIRM TODAY
Exactech Recall FAQ
- OPTETRAK® All-polyethylene PS Tibial Components
- OPTETRAK® CR Tibial Inserts
- OPTETRAK® CR Slope Tibial Inserts
- OPTETRAK® PS Tibial Inserts
- OPTETRAK® HI-FLEX® PS Tibial Inserts
- OPTETRAK Logic®
- OPTETRAK Logic® CR Slope Tibial Inserts
- OPTETRAK Logic® CRC Tibial Inserts
- OPTETRAK Logic® PS Tibial Inserts
- OPTETRAK Logic® PSC Tibial Inserts
- OPTETRAK Logic® CC Tibial Inserts
- TRULIANT® CR Slope Tibial Inserts
- TRULIANT® CRC Tibial Inserts
- TRULIANT® PS Tibial Inserts
- TRULIANT® PSC Tibial Inserts
- VANTAGE® VANTAGE® Fixed-Bearing Liner
After extensive testing, the company confirmed that most inserts manufactured since 2004 were packaged in out-of-specification vacuum bags that are oxygen resistant but do not contain a critical secondary barrier layer that further augments oxygen resistance.
The use of these non-conforming bags may enable increased oxygen diffusion to the insert, resulting in increased oxidation of the material relative to inserts packaged with an additional oxygen barrier. Oxidation can severely degrade the mechanical properties of the device and can lead to accelerated wear debris production, bone loss, and component fracture, all leading to revision surgery.
Our Firm will help you find the answers. The Firm has the experience, resources and dedication to take on difficult and emotional cases and help our clients obtain the justice for the wrong they have suffered.
Experience: Joe Lyon is an experienced Defective Medical Device Lawyer. The Lyon Firm has 20 years of experience and success representing individuals and plaintiffs in all fifty states, and in a variety of complex civil litigation matters. Defective device and recall lawsuits can be complex and require industry experts to determine the root cause of an accident or injury. Mr. Lyon has worked with experts nationwide to assist individuals understand why an injury occurred and what can be done to improve their lives in the future. Some cases may go to a jury trial, though many others can be settled out of court.
Resources/Dedication: Mr. Lyon has worked with experts in the fields of accident reconstruction, biomechanics, epidemiology, metallurgy, pharmacology, toxicology, human factors, workplace safety, life care planning, economics, and virtually every medical discipline in successfully representing Plaintiffs across numerous areas of law. The Lyon Firm is dedicated to building the strongest cases possible for clients and their critical interests.
Results: Mr. Lyon has obtained numerous seven and six figure results in medical device cases. He has litigated cases successfully against some of the largest companies in the world including: Johnson & Johnson, Biomet, Ethicon, Stryker, Coloplast, Smith & Nephew, American Medical Systems, Boston Scientific, Medtronic, Guidant, Bard, & Bayer.
A medical device is defective if it is unreasonably dangerous for its intended use. A legal cause of action can be based on several types of medical device product defects. Most jurisdictions have a version of one or more of these cause of actions:
(1) Manufacturing/ Construction Defect of the medical device:
(2) Defective design and/or formulation of the medical device:
(3) Failure to warn or inadequate warning or instruction associated with the medical device:
(4) Misrepresentation on the Safety or Efficacy of the Medical Device:
(5) Fraud Related to the Safety or Efficacy of the Medical Device
(6) Negligent Distribution or Testing of the Medical Device
- Hernia Mesh
- Breast Implants
- Transvaginal Mesh Implants
- Catheters
- Hip Replacements
- Knee Implants
- Ankle Replacements
- Shoulder Replacements
- Heater Coolers
- Bone Grafts
- Ventilators
- Defibrillators
- IUD
- IVC Filters
- Da Vinci Robot Surgery System
Medical Device Manufacturers
- Atrium Hernia Mesh
- Bard Hernia Mesh
- Bard IVC Filters
- Covidien Hernia Mesh
- Depuy Hip Implants
- Arthrex knee replacement
- Exactech knee replacement
- Exactech ankle replacement
- CareFusion ventilators
- Essure Birth Control System
- Ethicon hernia mesh
- Infuse Bone Grafts
- LivaNova Heater-Coolers
- Stryker Hip Implants
- Smith & Nephew Hip Systems
- Zimmer Biomet Shoulder Systems
- Wright Medical Hip Implants
Defective design medical device cases arise not because a mistake was made during the manufacturing process, but rather the original design of the product is unreasonably dangerous. A “risk-benefit analysis” is used to determine whether safer/less expensive alternative designs were available to the manufacturer (e.g. the metal on metal design, the porosity and stiffness of transvaginal mesh).
For these claims, the FDA Review process is critical on whether a pre-emption defense exists. For devices that went through the PMA (pre-market approval process), rather than the 510K process, the case must be evaluated very carefully for any parallel claims under state and federal regulations (e.g., failure to warn or failure to disclose adverse events) that can advance the case forward. Without a careful legal analysis, the case may be subject to dismissal even where the medical device design proves to be defective.
Risks: The following factors are considered under Ohio law when determining the risks associated with the design of a medical device: (1) the magnitude of the risk of injury; (2) ordinary consumer awareness of the risk for injury; (3) the likelihood of causing injury; (4) the violation of a private or public standard; and (5) the consumer’s expectation of the performance of the product and level of danger. Ohio Revised Code 2307.5 (B) Product Defective in Design or Formulation. These factors are considered in most other jurisdictions.
Benefits: The following factors are considered under Ohio law when determining the benefits associated with a medical device: (1) the utility of the product; (2) availability of an alternative design; (3) the magnitude of risks associated with an alternative design. Ohio Revised Code 2307.5 (c) These factors are considered in most other jurisdictions.
Defenses for Defective Design: (1) a pharmaceutical drug or medical device is not defective by design if it contains an adequate warning of an unavoidably unsafe aspect of the pharmaceutical or medical device; (2) the dangerous aspect is inherent to the product, recognizable, and cannot be eliminated without compromising the product’s usefulness; (3) a lack of a feasible alternative design. 2307.75 (d)(e)(f).
A manufacturing defect claim arises where the medical device is released from the factory in a manner that deviates from the intended design or specifications.
Most manufacturing defect cases are based on a products deviation from the intended specification, formula, performance standards, or design model. In such cases, it may be easy to determine the product did not comply with the intended design. In defective medical device cases, the manufacturing defect is often related to performance standards (e.g., the metal does not meet the strength specification or has microscopic fractures–these defects require an examination by a qualified metallurgist who can examine the medical device).
As a result of the deviation, the product enters the market in an unreasonably dangerous condition and the consumer is exposed to or purchases a product that is defective. Any personal injuries or economic loss that arise from the the defect are compensable under product liability law.
The product may be recalled as a specific lot is identified as being non-compliant and defective. A product may be defective in manufacture or construction, materials and assembly, and a manufacturer or distributor may be subject to strict liability, even though it exercised all possible care.
All medical device products come with warnings and instructions for use that are provided to the physician. If the company does not provide the physician with all the relevant safety data so that the physician adequately understand the risks and benefits of the product, then the medical device is defective.
The physician must be in a position to pass the appropriate safety data onto the patient, so the patient can make an informed decision on whether they wish to have the medical device implanted. Patient informed consent is critical because many devices are designed to be permanent.
Litigation arises where there is evidence the medical device manufacturer failed to timely update a warning in light of new data or simply ignored the risk and failed to conduct sufficient research to identify and then disclose the risk.
In determining whether a medical device is defective due to inadequate warning or instruction, evidence must be presented to prove:
- The medical device manufacturer knew, or in the exercise of reasonable care, should have known about a risk
- A reasonable medical device manufacturer would have provided a warning of the risk
- The medical device manufacturer failed to provide the warning
- The patient was injured due to a lack of warning. The same elements apply whether the claim is based on a warning present during the marketing or post-sale warnings.
Defenses to Failure to Warn Claims: (1) the risk was open and obvious or a matter of common knowledge; and (2) in cases of a pharmaceutical drug or medical device, the warning was provided to the prescribing physician (“Learned Intermediary Doctrine”).
Design and manufacturing defects result in medical device product recalls each year in the United States, initiated by federal safety agencies.
The foregoing listed government agencies may initiate recalls of dangerous products but do not provide remedies or compensation for damages where an individual is injured due to the defective product. Only litigation can be utilized to compensate patients injured by a defective medical device.
Learn about Medical Device Approval Processes
The Lyon Firm aggressively, professionally, and passionately advocates for injured individuals and families against companies due to a defective product or recalled product to obtain just compensation under the law.
IN RE: DEPUY ASR
METAL ON METAL HIP IMPLANTS
Nationwide Consolidation in U.S. District Court, Northern District of Ohio. The Depuy ASR Hip design was plagued with problems related to premature loosening and metallosis, caused by chromium and cobalt entering the blood stream. The resulting injuries were a result of the metal-on-metal friction inherent with the design of the product. Depuy recalled the ASR and entered into a global settlement valued at $2.5 Billion to cover an estimated 8,000 patients. The Lyon Firm represented several clients in the original settlement and continues to be active in this litigation. The settlement funds provided compensation for the revision surgeries, including compensating for medical costs, lost wages, and pain and suffering.
IN RE: BIOMET MSA MAGNUM
METAL ON METAL HIP IMPLANTS
Nationwide Consolidation in U.S District Court, Northern District of Indiana. Similar to the Depuy ASR, the Biomet metal-on-metal hip design allegedly contributed to premature hip failures and metallosis due to the metal friction. Biomet entered into a global settlement valued at $56 million to cover a few thousand claims. The Lyon Firm represented several plaintiffs in the original settlement and continues to be active in this litigation.
Medical Device Information Center
-
-
Answer a few general questions.
-
A member of our legal team will review your case.
-
We will determine, together with you, what makes sense for the next step for you and your family to take.
-